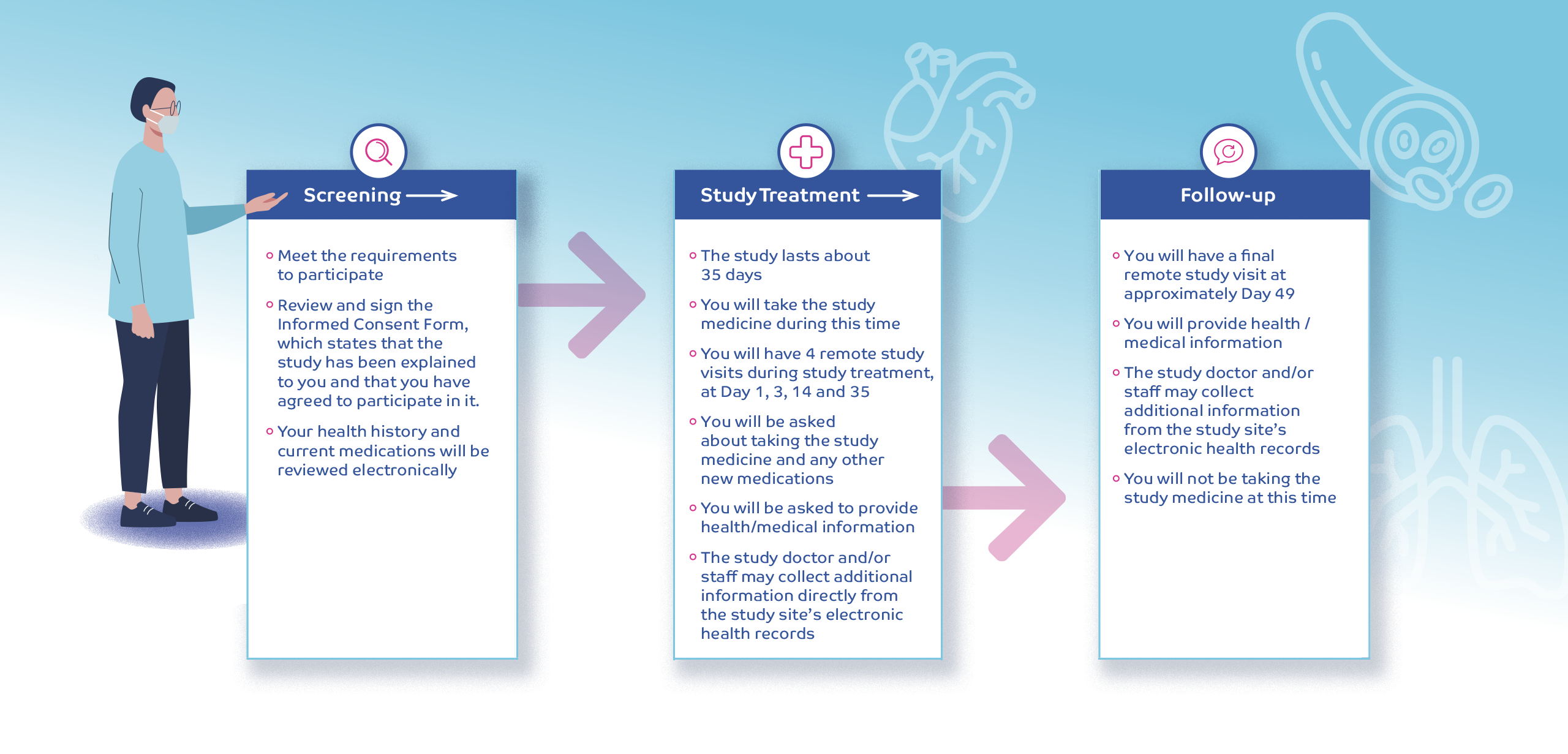
THE CLINICAL TRIAL
Coronavirus disease (COVID-19) is an infectious disease caused by the SARS-CoV-2 virus. While the disease caused mild-to moderate respiratory symptoms in many, some experienced severe illness with serious complications, such as blood clots that could lead to heart attack stroke, hospitalization, or even death. The purpose of the PREVENT-HD Trial was to evaluate if an investigational drug could prevent these complications.
THE PROJECT
The pandemic was an anxiety-filled two years that turned our routines upside down. Misinformation and disinformation were running rampant, causing an unprecedented distrust in the scientific community. There was no vaccine available at this time, and very little was known about COVID-19 symptoms and complications. Our approach focused on breaking through to patients during this stressful time and educating patients at the ever-changing point of care.